Slide 01 — Overview

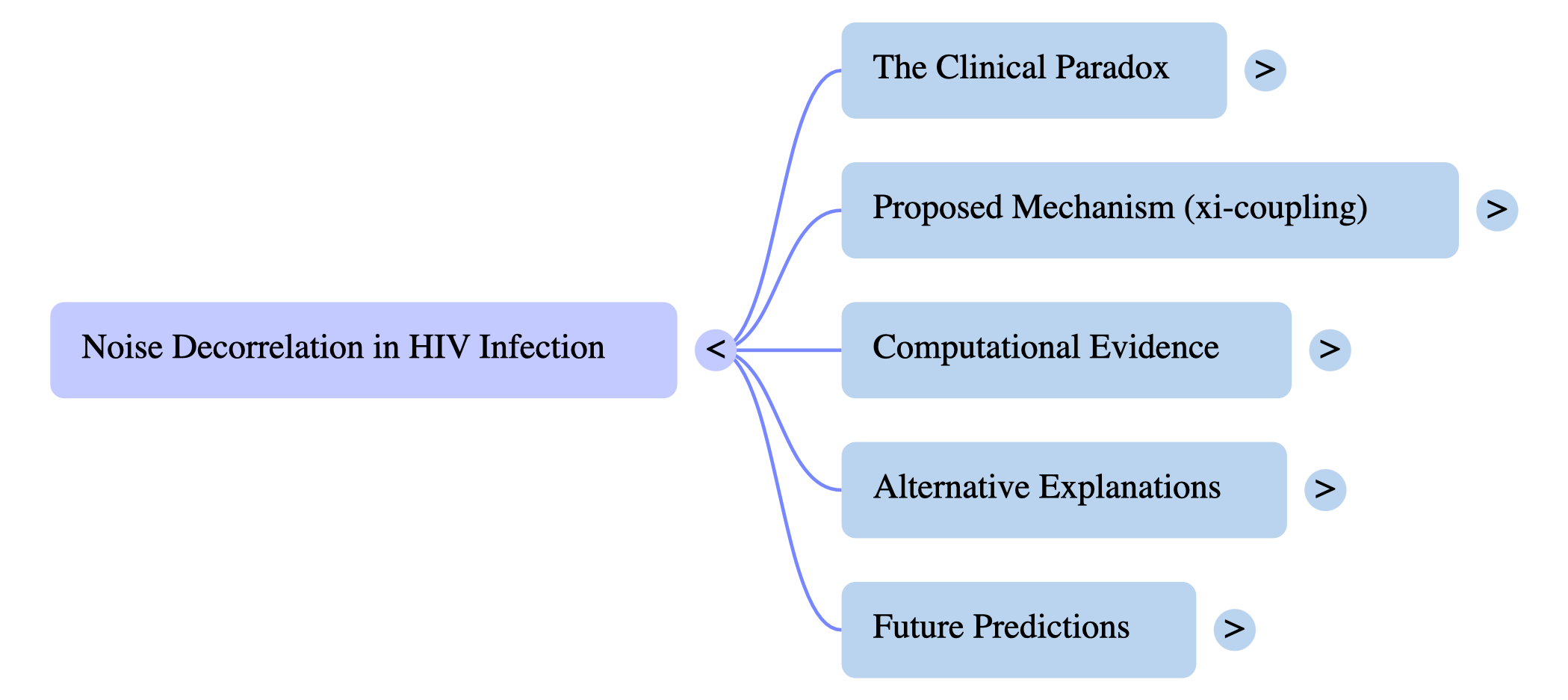
Toward solving the 35-year-old paradox of HIV neurometabolic outcomes. During acute HIV, viral loads reach 107 copies/mL with massive inflammation — yet 80–93% of patients remain neurocognitively asymptomatic. In the chronic phase, viral load is suppressed, yet neurons progressively die.
Slide 02 — The Evolutionary Constraint

Neurons cannot divide. Neuronal loss is permanent. Unlike liver or skin cells, mature CNS neurons are non-renewable. Evolution faces a critical problem: how to protect irreplaceable tissue during a massive inflammatory event.
Slide 03 — The Clinical Paradox
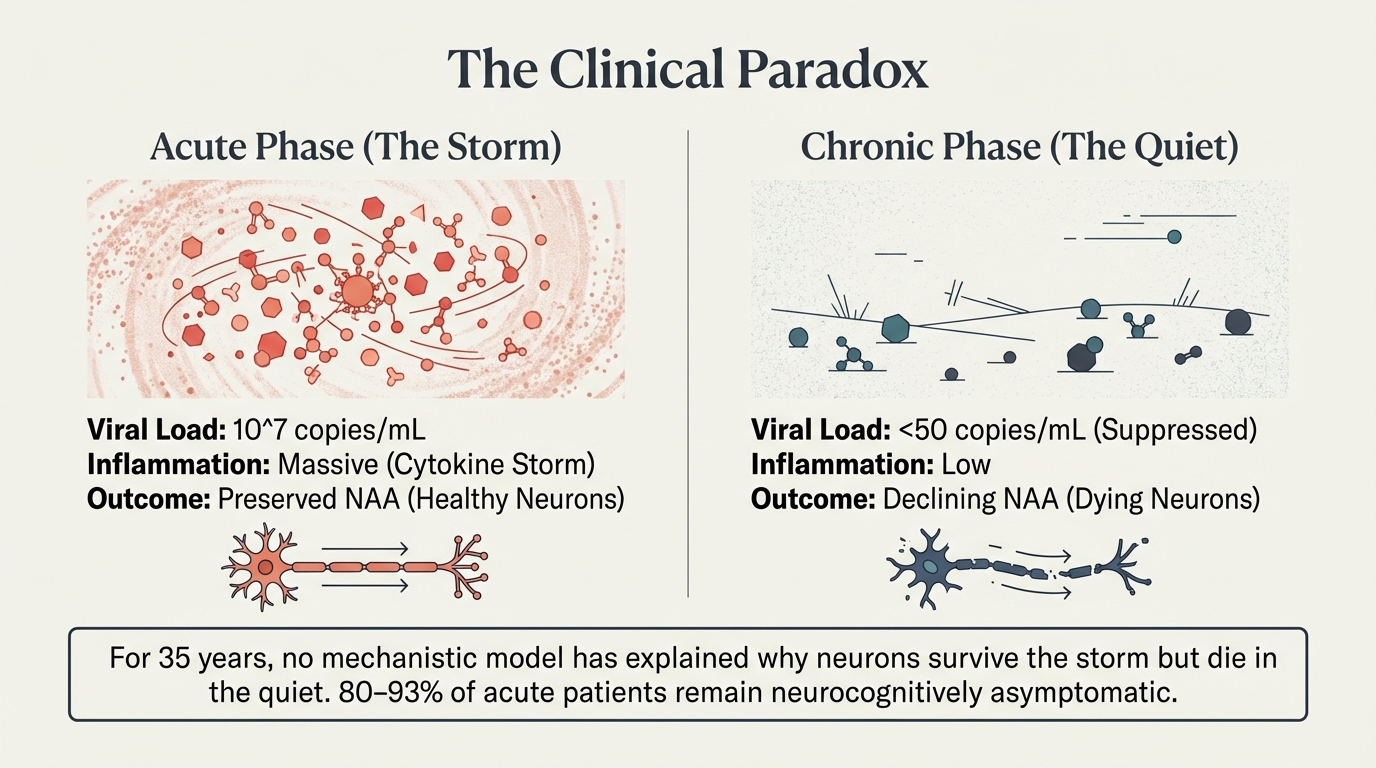
The storm preserves. The quiet destroys. Acute phase: viral load at 10^7, massive cytokine storm, yet NAA (neuronal integrity marker) is preserved. Chronic phase: viral load <50, low inflammation, yet NAA declines. For 35 years, no mechanistic model has explained this.
Slide 04 — Structure, Not Amplitude
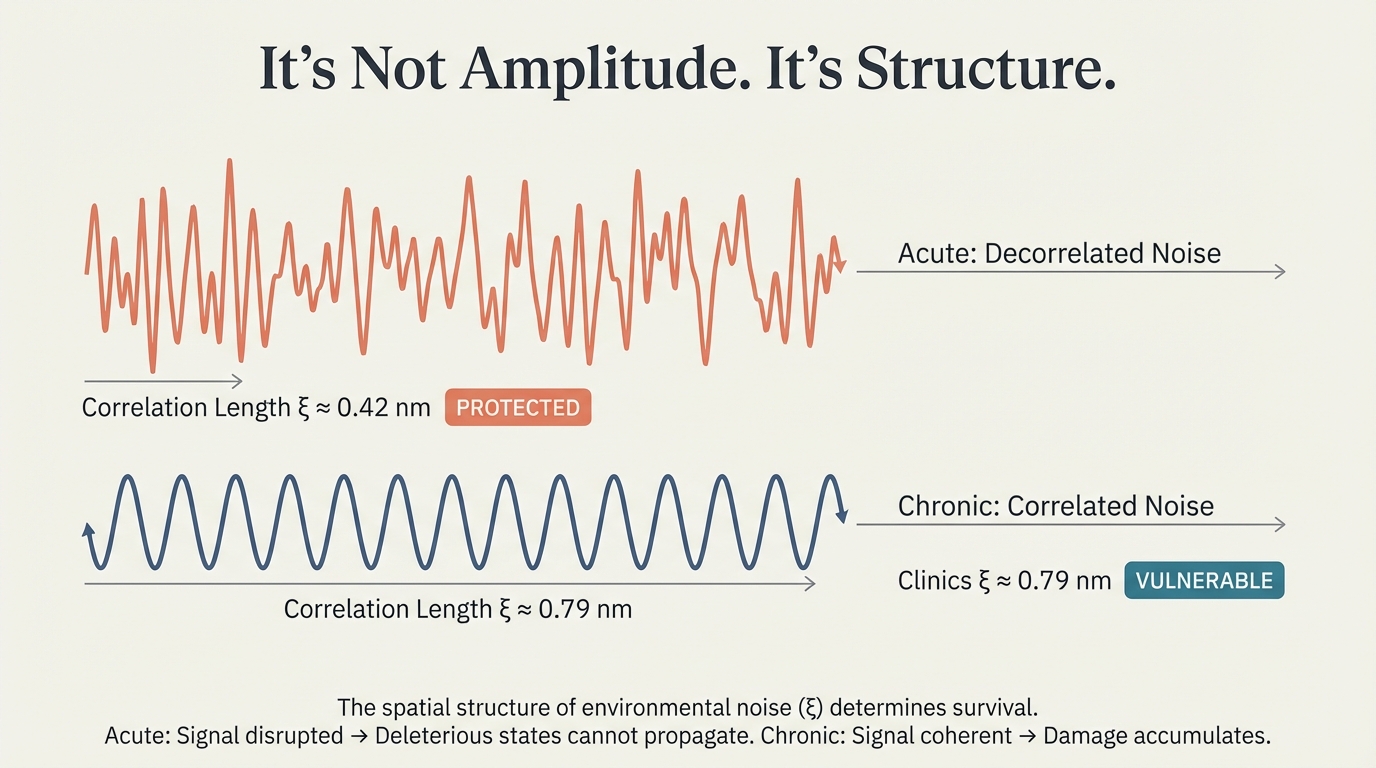
The spatial structure of environmental noise determines survival. Acute: decorrelated noise (ξ ≈ 0.42 nm) — signal disrupted, deleterious states cannot propagate. PROTECTED. Chronic: correlated noise (ξ ≈ 0.79 nm) — signal coherent, damage accumulates. VULNERABLE.
Slide 05 — Quantum Protection

Like noise-canceling headphones for metabolic stress. Chronic correlated noise allows coherent excitation trapping at NAT8L, causing damage. Acute decorrelated noise enables noise-assisted decoherence, scattering deleterious energy. Analogous to photosynthetic complexes that exploit noise to prevent coherent excitation trapping.
Slide 06 — Evolutionary Optimization
The perfect solution for acute stress in non-renewable tissue. Speed: instant response, no gene expression lag. Cost: metabolically cheap, uses environmental noise. Reversibility: transient, turns off when threat passes. It uses the physics of inflammation itself.
Slide 07 — Bayesian Methodology
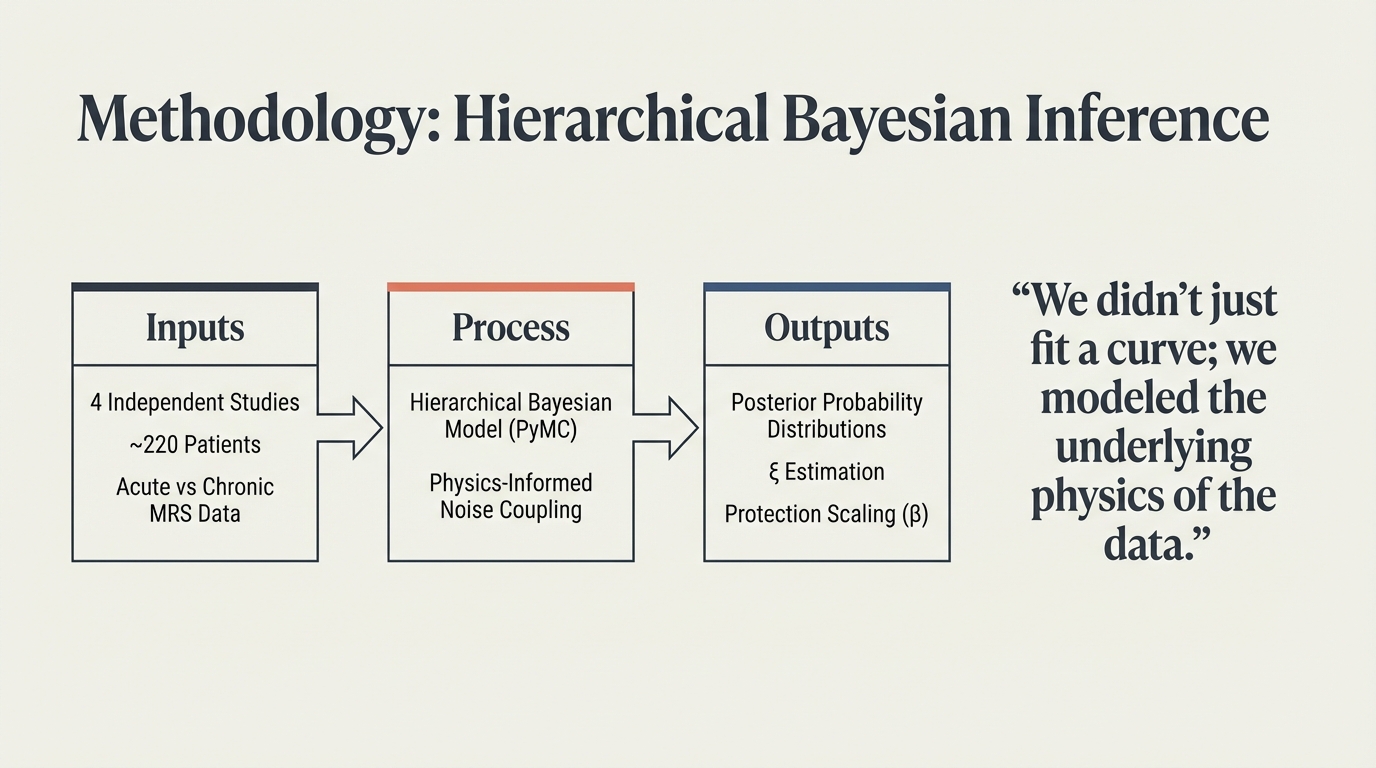
“We didn’t just fit a curve; we modeled the underlying physics of the data.” 4 independent studies, ~220 patients, acute vs chronic MRS data. Hierarchical Bayesian model (PyMC) with physics-informed noise coupling yields posterior distributions, ξ estimation, and protection scaling (β).
Slide 08 — Distinct Physical Regimes
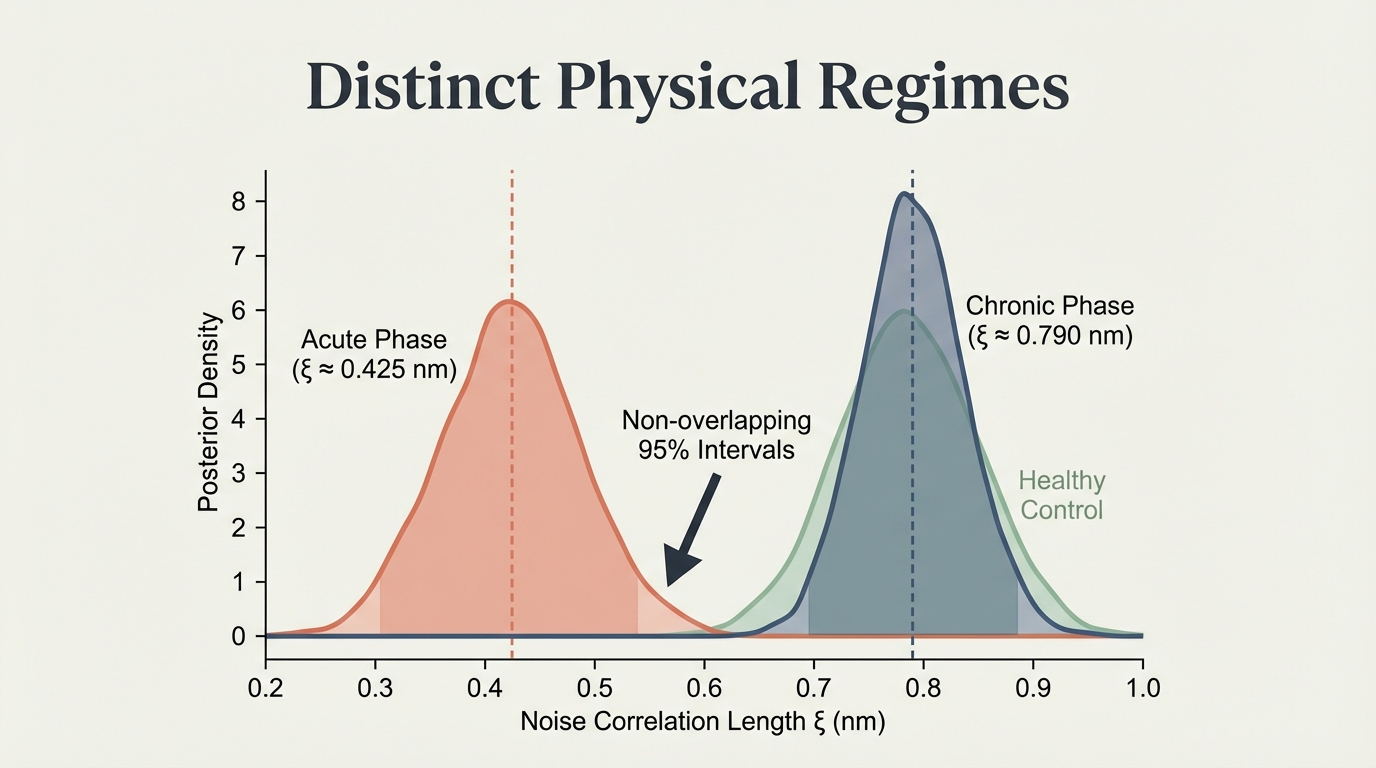
Acute and chronic HIV occupy completely distinct noise regimes. Acute: ξ ≈ 0.425 nm. Chronic: ξ ≈ 0.790 nm. The 95% posterior intervals do not overlap. Healthy controls cluster with the chronic phase, confirming that the acute state is the anomaly — the protected state.
Slide 09 — Statistical Decisiveness

This is not noise; it is a signal. Bayes Factor >1000 (decisive evidence). Cohen’s d = 5.63 (massive effect size). P(ξacute < ξchronic) >99.9%. In clinical statistics, effect sizes of this magnitude are exceptionally rare.
Slide 10 — Superlinear Scaling
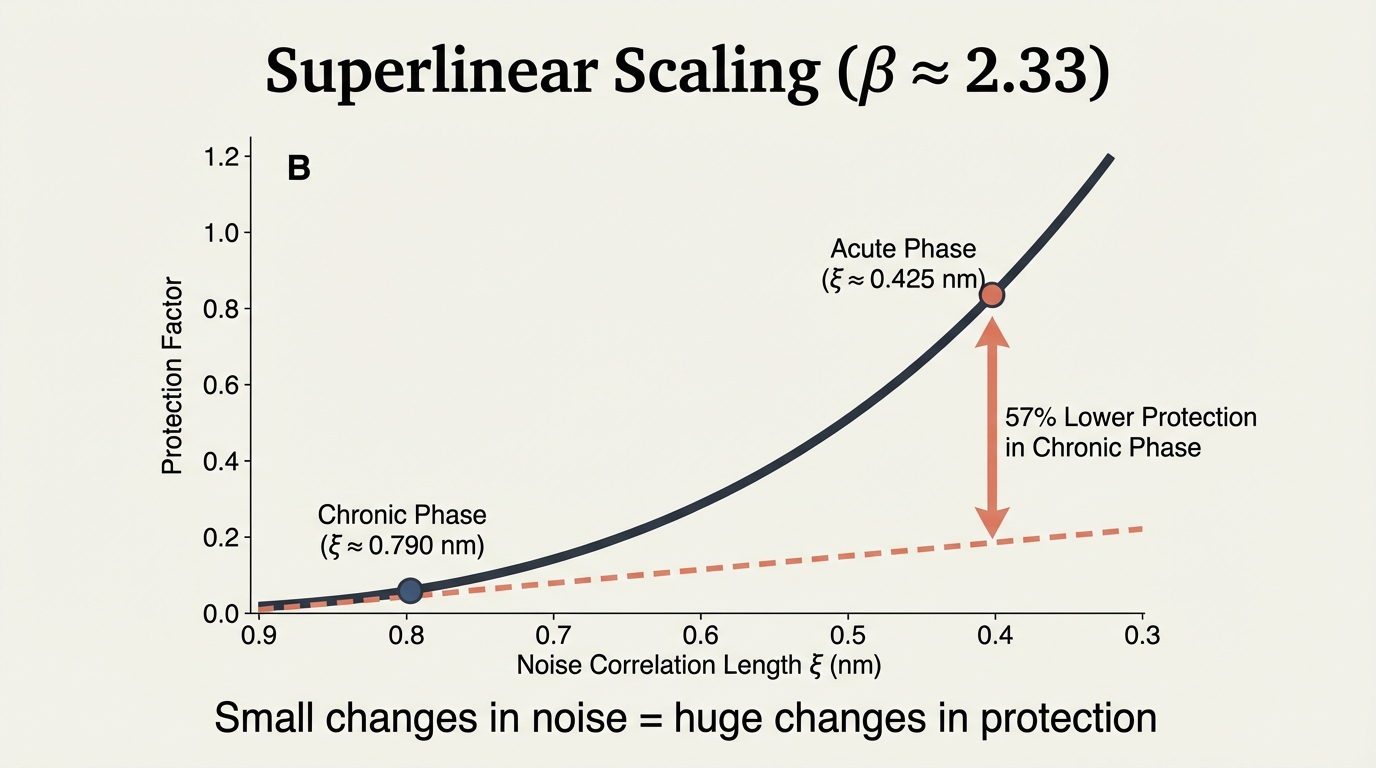
Small changes in noise produce huge changes in protection. Protection scaling exponent β ≈ 2.33 is superlinear. Chronic phase shows 57% lower protection. This nonlinear amplification explains why even modest noise structure changes have dramatic neurological consequences.
Slide 11 — Validation
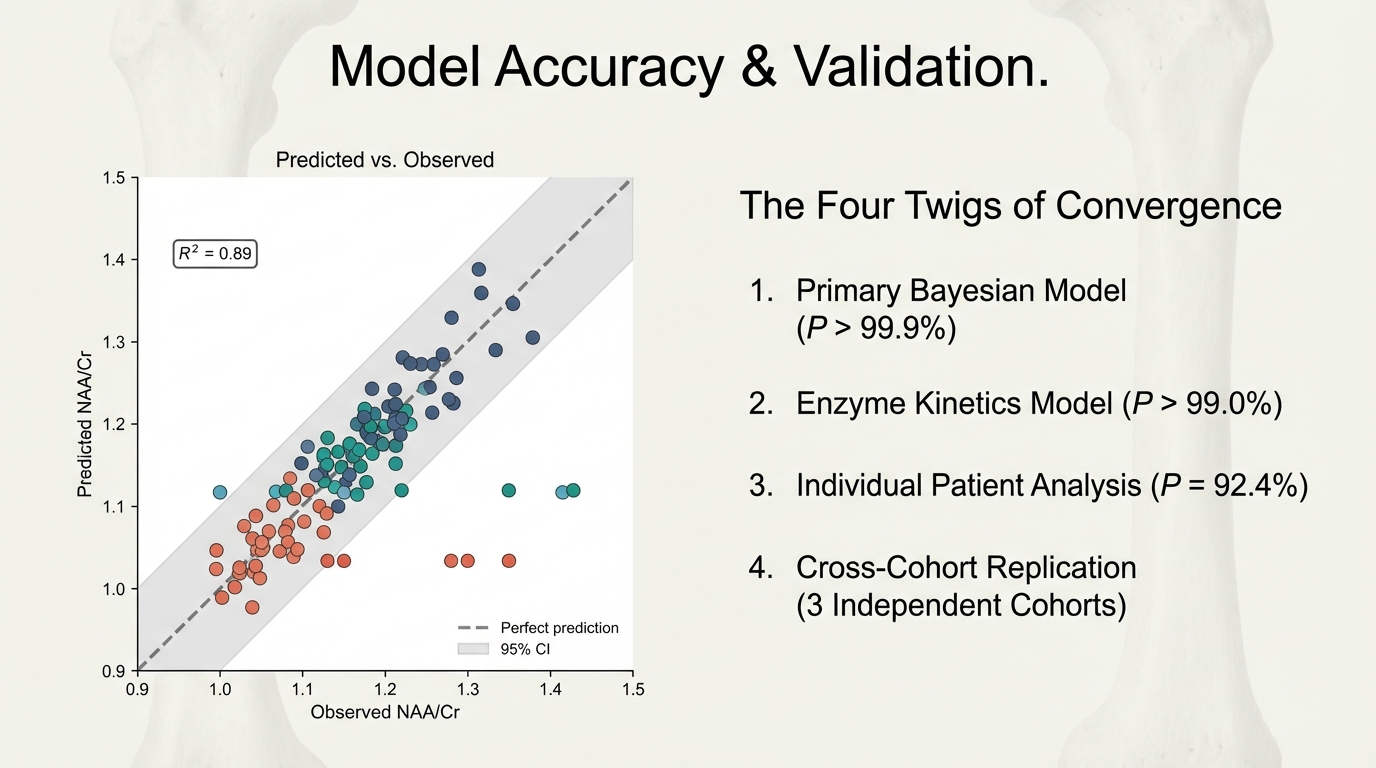
Four independent lines of evidence converge. Primary Bayesian model (P > 99.9%). Enzyme kinetics model (P > 99.0%). Individual patient analysis (P = 92.4%). Cross-cohort replication (3 independent cohorts). Predicted vs. observed NAA/Cr: R² = 0.89.
Slide 12 — Phylogenetic Protection
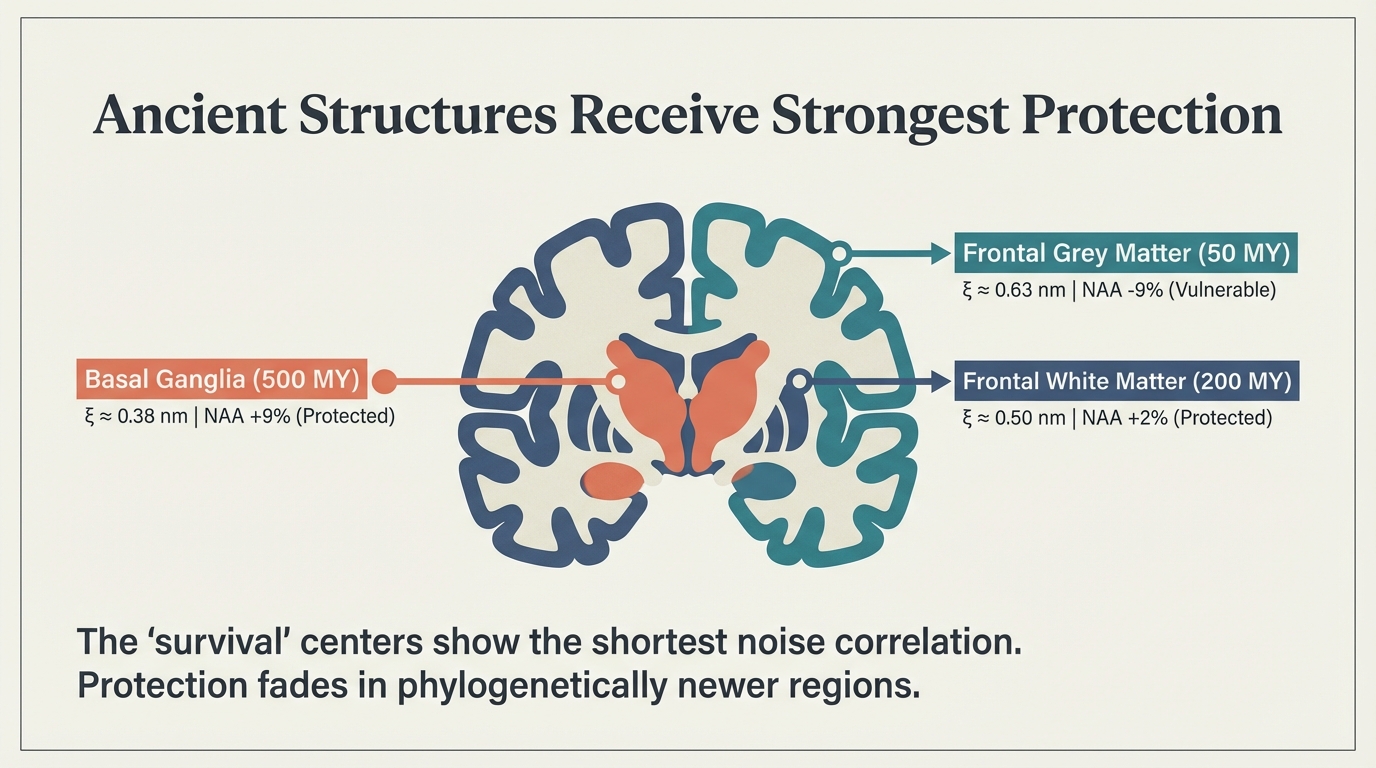
The “survival centers” show the shortest noise correlation. Basal ganglia (500 MY): ξ ≈ 0.38 nm, NAA +9%. Frontal white matter (200 MY): ξ ≈ 0.50 nm, NAA +2%. Frontal grey matter (50 MY): ξ ≈ 0.63 nm, NAA -9%. Protection fades in phylogenetically newer regions.
Slide 13 — The Window of Opportunity
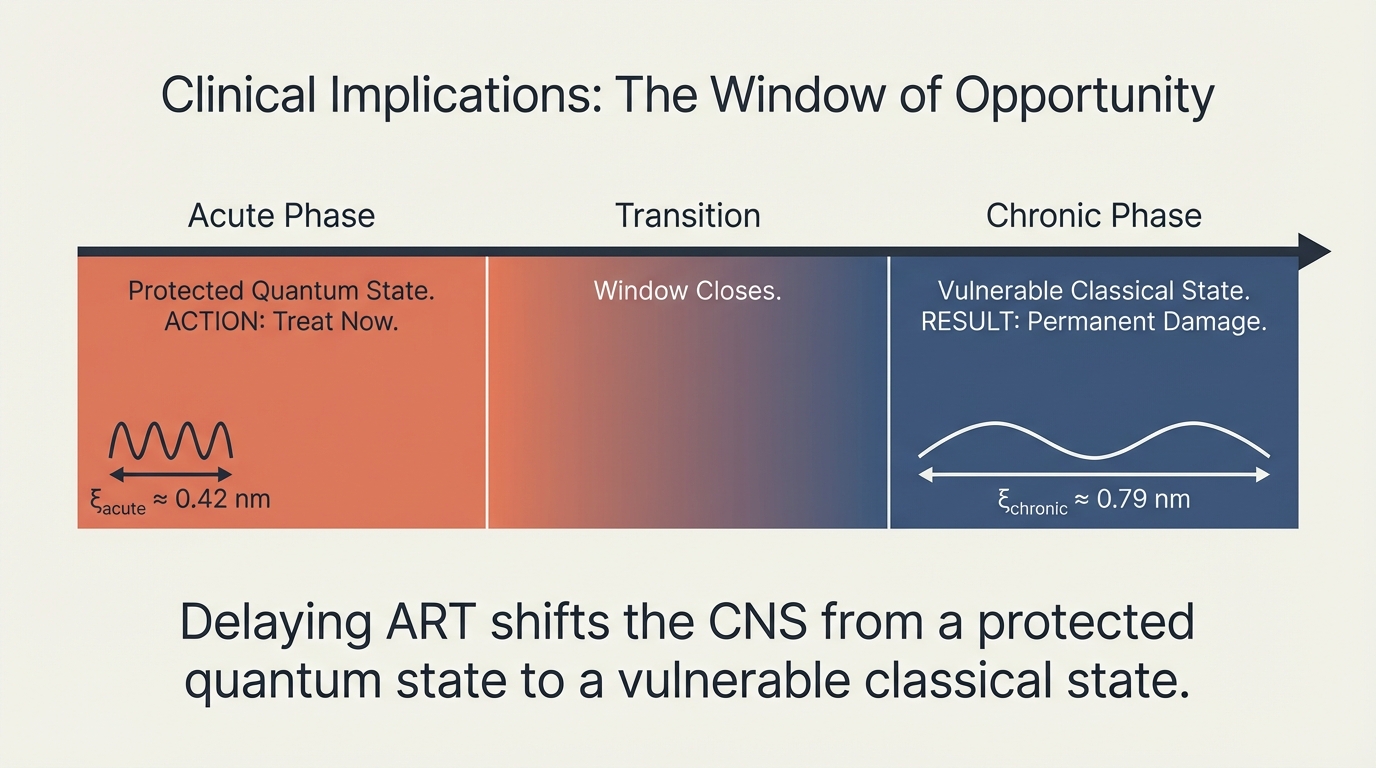
Delaying ART shifts the CNS from a protected quantum state to a vulnerable classical state. Acute (ξ ≈ 0.42 nm): protected — treat now. Transition: window closes. Chronic (ξ ≈ 0.79 nm): vulnerable — permanent damage. Early ART initiation is neuroprotective.
Slide 14 — Testable Framework
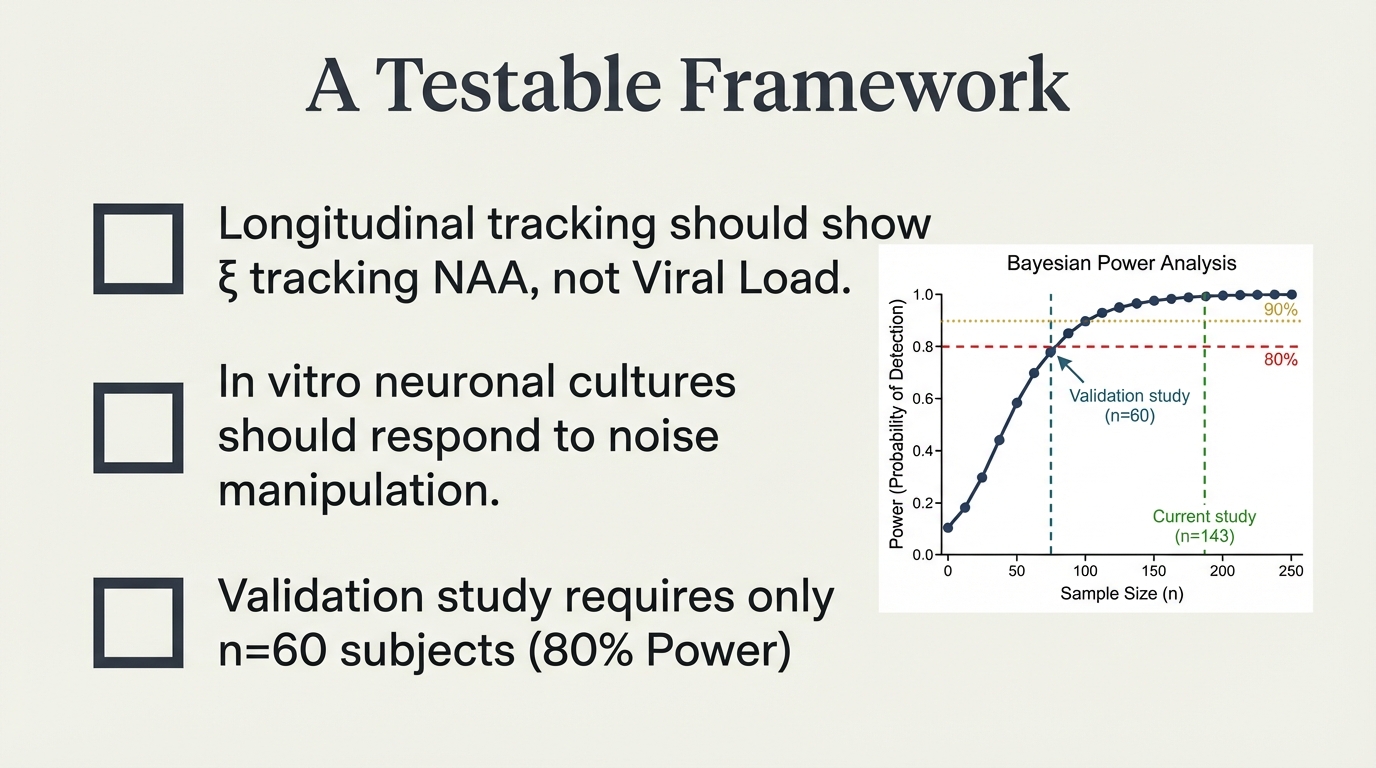
Three falsifiable predictions. Longitudinal tracking should show ξ tracking NAA, not viral load. In vitro neuronal cultures should respond to noise manipulation. Validation requires only n=60 subjects for 80% power — the effect size is so large that modest samples suffice.
Slide 15 — Proposed Solution
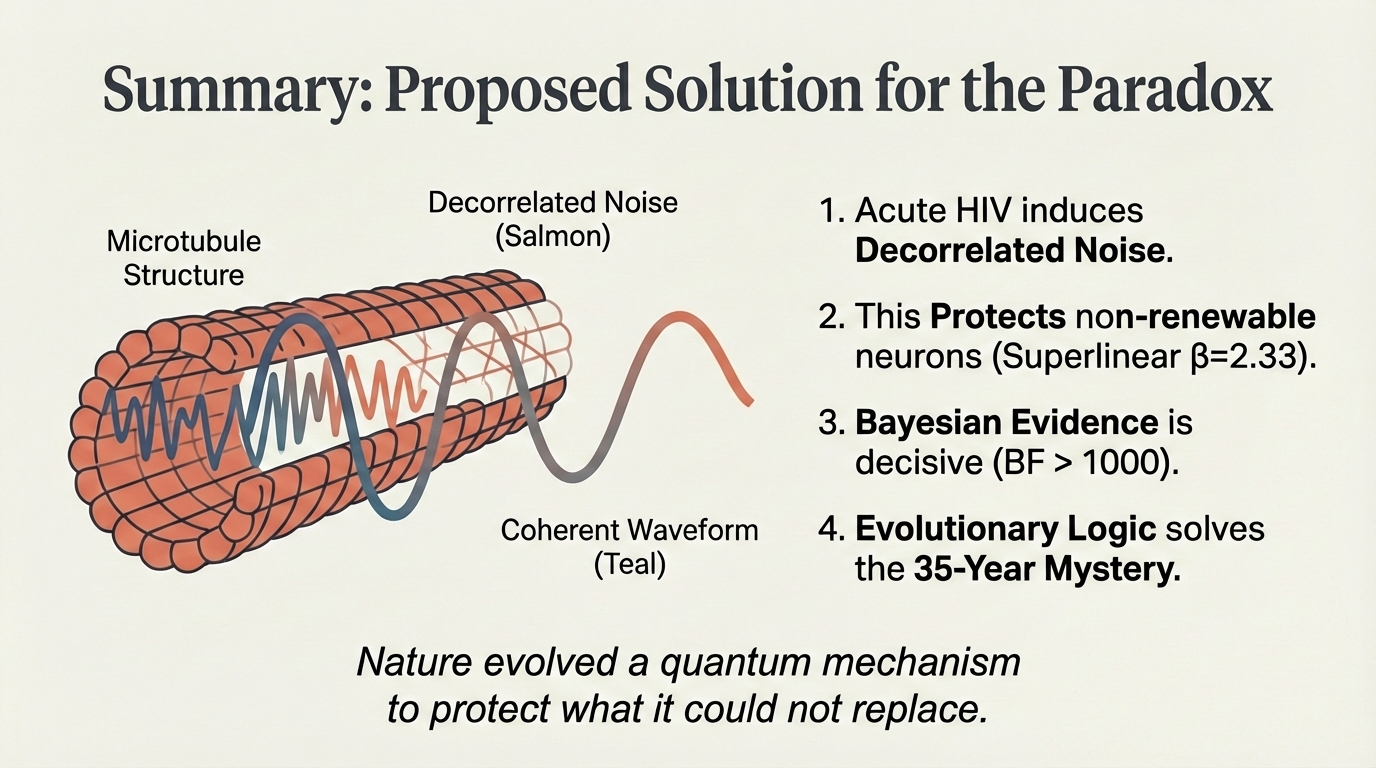
Nature evolved a quantum mechanism to protect what it could not replace. 1. Acute HIV induces decorrelated noise. 2. This protects non-renewable neurons (superlinear β=2.33). 3. Bayesian evidence is decisive (BF > 1000). 4. Evolutionary logic solves the 35-year mystery.